General Practice and HBOT
Hyperbaric Oxygenation Effects on Blood Flow
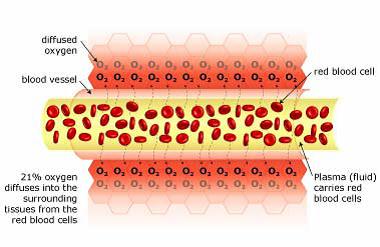
Normal blood flow
There is 21% oxygen in the air that we breathe, and our lungs transfer this oxygen to our red blood cells (via hemoglobin). These oxygen-filled red blood cells are carried around the body by the plasma (fluid), which travels through the blood vessels. The oxygen diffuses into the surrounding tissue ensuring that it is delivered to where it is needed most.
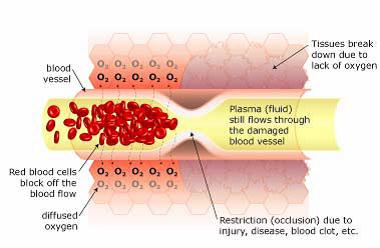
Restricted blood flow (ischemia) causes Hypoxia
When there is a restriction (occlusion) in blood flow due to surgery, illness, or injury, the red blood cells block the blood vessel and are unable to transfer oxygen to the cells on the other side of the occlusion. This causes swelling and starves the area of oxygen, causing hypoxia (a lack of oxygen); when this occurs the tissue begins to break down. Hypoxia triggers ‘apoptosis’ (programmed cellular degeneration – clumping and clustering of damaged nerve cells surrounded by healthy neuronal tracts).
Apoptosis modifies the expression of plasticity (the ability of the body to repair). Apoptotic bodies and altered DNA fragmentations are observed in the avascular ischemic region with increased inhibitory biochemical factors (proteins) released into the damaged parts of the brain and spinal cord causing further deterioration. Apoptosis has been identified in all neurodegenerative disorders including brain and spinal cord injury. Apoptosis fosters the cycle of continued dysfunction, degeneration and ultimate neuronal death.
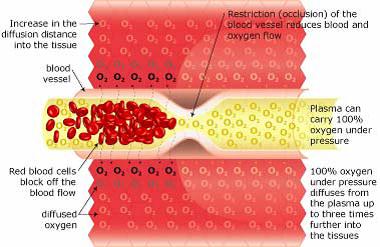
Hyperbaric Oxygenation
Breathing 100% oxygen under pressure causes the oxygen to diffuse into the blood plasma. This oxygen-rich plasma is able to travel past the restriction, diffusing up to 3 times further into the tissue. The pressurized environment helps to reduce swelling and discomfort, while providing the body with at least 10-15 times its normal supply of oxygen to help repair tissue damaged by the original occlusion or subsequent hypoxic condition. Hyperbaric Oxygenation (HBOT) directly increases the saturation of tissue oxygenation, slowing and reversing hypoxic induced apoptosis - restoring blood supply to the compromised region by the development of new capillary networks (neovascularization) enabling the body to alter the course and impact of the disease process.
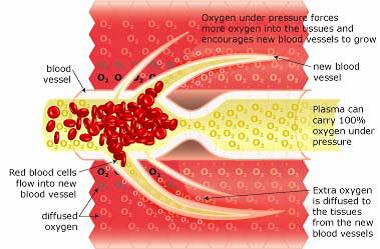
Neurovascular Regeneration
HBOT mobilizes the body’s circulating stem cells. American Journal Physiology - Heart and Circulatory Physiology (Nov 05)] reports a single 2-hour exposure to HBOT at 2 ATA doubles circulating CD34+ progenitor stem cells (primordial cells targeted to salvage and restore damaged structures); and at approx. 40-hours of HBOT; circulating CD34+ cells increases eight fold (800%).
Images by Malcolm R Hooper - HyperMED Australia
Case Studies
HBOT and Fibromyalgia
Fibromyalgia is characterized by longstanding multifocal pain with generalized allodynia/ hyperalgesia. We conducted a randomized controlled study to evaluate the effect of hyperbaric oxygen therapy in FMS (HBO group: n = 26; control group: n = 24). Tender points and pain threshold were assessed before, and after the first and fifteenth sessions of therapy. Pain was also scored on a visual analogue scale (VAS). There was a significant reduction in tender points and VAS scores and significant increase in pain threshold of the HBO group after the first and the fifteenth therapy sessions. There was also a significant difference between the HBO and control groups for all parameters except the VAS scores after the first session. We conclude that HBO therapy has an important role in managing FMS.
HIV-Related Fatigue and HBO
Hyperbaric Oxygen Therapy (HBO) is being evaluated to determine the effectiveness in relieving HIV-related fatigue and determine the clinical and immunological effects on HIV-infected individuals.
Twenty-five patients have been enrolled over a two-year period. Admission criteria include chief complaint of fatigue, seropositivity as confirmed by ELISA and Western Blot for HIV infection, 500 CD4 cells or less, and absence of active opportunistic infections which might compromise pulmonary or neurologic function. Twenty patients received 2 ATA, for 90 minutes, three times per week. Five patients voluntarily received surface air at the same parameters to control for a placebo effect, and then received 100% oxygen; all patients were treated simultaneously in the HBO chamber. Laboratory, clinical assessment, and Karnofsky Performance Scores were completed monthly on all patients.
The results show that all patients indicated relief of debilitating fatigue within two weeks. Karnofsky Scores improved 10% to 30% within one month. Weights for all patients remained stable or increased; CD4, Hemoglobin, and Hematocrit counts remained stable and/or increased. Tumor Necrosis Factor decreased in 13 patients. P-24 antigens remained non-reactive or decreased if reactive. All patients returned to their six-month Karnofsky Score, which indicated significant improvement in their fatigue levels. Twenty-three of the twenty-five patients have shown no clinical disease progression; 80% of patients enrolled in the study have 50 CD4 cells or less. Further, HBO appeared to relieve pain associated with peripheral neuropathy in two patients and was a beneficial adjunct to the treatment of mild PCP; the investigators have established protocols to further investigate these findings.
The conclusion is that HBO appears to be effective in relieving HIV-related fatigue and improving the quality of life of individuals with HIV/AIDS. Preliminary research indicates a probable correlation with an increase in length of employment capability, delayed disability requirement, and reduction in number and length of hospitalizations by incorporating HBO as a component in the medical management of HIV disease. The investigators believe HBO may relieve fatigue by inhibiting the activity of certain cytokines, enhancing red blood cell production, and enhancing the ability of HIV-impaired monocytes to absorb and utilize oxygen.
Reillo, M, R.N., B.S.N., Myers, R., M.D., HBO Staff, MIEMSS, Baltimore, Maryland, USA
RSD and HBOT as an adjunct to Sympathectomy.
Thomas PS; Hakim TS; Trang LQ; Hosain SI; Camporesi EM,Department of Anesthesiology, State University of New York Health Science Center, Syracuse 13210, USA. hschosp.umag.thomasp. Anesth Analg, 88(1):67-71 1999 Jan
The aim of this study was to determine whether regional sympathectomy attenuates this vasoconstriction response and thus improves oxygen delivery. In a double-blinded manner, healthy volunteers were subjected to HBO2 in a monoplace chamber on two occasions separated by at least 1 wk. Transcutaneous oxygen (tcPO2) and carbon dioxide (tcPCO2) on the forearm were monitored continuously, and blood flow in the axillary artery was measured using angiodynography before and after exposure to HBO2. During one visit, each volunteer received a sympathetic block to the upper extremity by an injection of lidocaine into the brachial plexus at the axilla. During a second visit, the volunteer received a placebo injection of isotonic sodium chloride solution into the brachial plexus of the same side. Skin temperature was recorded on the back of the hand.
All subjects exhibited a small but significant increase in skin temperature (2.5%) and in upper limb blood flow (23%) (P < 0.05%) after sympathectomy, but not after isotonic sodium chloride solution injection. Sympathectomy increased tcPO2 marginally while in room air.
However, during HBO2, tcPO2 was substantially and significantly higher (409.8+/-98.8 mm Hg) after sympathectomy compared with that after isotonic sodium chloride solution injection (171.3+/-38.1 mm Hg). tcPCO2 did not change significantly after sympathectomy or during HBO2. Thus, sympathectomy presumably improved oxygen delivery by preventing vasoconstriction during hyperoxia. The results suggest that sympathectomy may be a useful adjunct to HBO2 therapy in patients in whom vascular resistance is increased because of sympathetic tone or hyperoxia.
IMPLICATIONS: Sympathetic nerve block of the extremities markedly enhances tissue oxygen delivery during hyperbaric oxygen treatment. Sympathectomy may be a beneficial adjunct treatment to hyperbaric oxygen in peripheral vascular insufficiency.
Hyperbaric Oxygen Therapy Process Indications
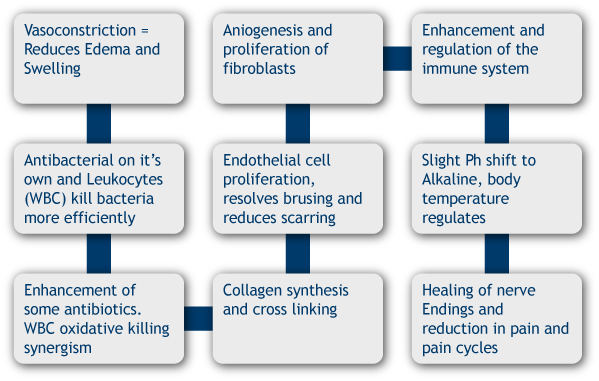
Many patients, when following closely the prescribed protocol for Hyperbaric Oxygen Therapy notice the following cumulative benefits from the oxygen:
To see what conditions related to your medical field are treatable with Hyperbarics click on the link below.
Contraindications
To see a PDF file detailing reasons why you might not prescribe HBOT, click below.
Contraindications (PDF)